FEATURE
Accelerating Collaborative Sustainability in the Tapes Industry
Evaluating Modifications in Aspartic Systems on Viscoelastic Behavior
Opportunities to increase market share will increase when companies are pre-positioned for growth in a resilient and scalable way.
Evaluating Modifications in Aspartic Systems on Viscoelastic Behavior
By Lisa Anderson, Founder and President, LMA Consulting Group
By John Winfrey, Director, Systems & Applications Technology, Pflaumer Brothers, Ewing, New Jersey
Formulation choices in aspartic chemistry directly influence stiffness, flexibility, and thermal performance.
Introduction
For thirty 30 years, aspartic ester chemistry has been employed to offer coatings, adhesives, sealants, and elastomers with unique properties to include one-coat application at thicknesses to optimize long-term durability, color stability to UV exposure, high abrasion resistance, and VOCs, to name four. Over the years, materials based on this technology have been modified to impart varying characteristics. Each alteration impacts the final product, which requires thorough testing to ensure the system's suitability for a specified application. This paper shares laboratory results of employing materials within a polyaspartic coating with varying structures and using Dynamic Mechanical Analysis (DMA) to understand the impact on viscoelastic properties when implementing formula variables.
DMA is a technique for evaluating mechanical properties such as stiffness, flexibility, and thermal stability by assessing the viscoelastic behavior of materials under controlled conditions. While the method can be utilized for various materials, this paper focuses on compositional influences on aspartic-based chemistry materials.
The commercialization of polyaspartic coatings began in the early 1990s, primarily driven by the need for more environmentally friendly alternatives to traditional coatings. Specifically, polyaspartic esters could reduce the viscosity of polyurethane-based materials, reducing the solvent demands of the coating material and resulting in lower VOCs, a driving demand from regulatory agencies to minimize pollutants derived from coatings during that period and continuing to the present. Polyaspartic esters became highly valued for their ability to cure rapidly without needing fast-drying solvents, which are traditionally employed in other types of coatings based on alternative chemistry building blocks.
After 30 years of working with this unique chemistry, researchers have found ways to adjust systems based on polyaspartic chemistry — slowing reactivity, adding flexibility, etc. — to accommodate the requirements for specific needs. With this work, the DMA can bring insight into how these alterations impact the final coating to understand the benefits or weaknesses of the modifications.
Experimental
DMA is a technique for studying the mechanical properties of materials as a function of temperature, time, frequency, and applied stress. It involves using a sinusoidal, oscillatory force to measure a sample's response. DMA provides detailed information on a material's viscoelastic behavior, which refers to its combined elastic (solid-like or shape recovery response) and viscous (liquid-like or dispersing of energy response) properties.[1] The properties measured by DMA include [2]:
- Storage Modulus (E'): This represents the elastic component of the material, indicating its ability to store energy. It correlates with the material's stiffness and rigidity. Higher storage modulus values indicate a more rigid material.
- Loss Modulus (E"): This represents the viscous component, indicating the material's ability to dissipate energy. A higher loss modulus typically indicates better energy dissipation. For coatings, a high value means the material’s ability to resist damage from forces such as improvements in impact resistance.
- Tan Delta (δ): The ratio of loss modulus to storage modulus provides insights into the material's damping characteristics (resistance to vibrations) and energy dissipation. Understanding flexibility through DMA can ensure long-term performance for materials that must endure deformation for specified applications.
- Glass Transition Temperature (Tg): The temperature at which the material transitions from a rigid, glassy state to a more flexible, rubbery state. The Tg is a key indicator of a material's thermal behavior. Higher Tg materials exhibit better thermal stability. Understanding a material’s Tg helps predict its performance under various thermal conditions.
It should be noted that glass transitions occur over a temperature range, not a single temperature value for polymeric materials. While the peak value obtained for tan delta is traditionally reported in literature, it best represents permanent deformation. Additionally, while DMA provides glass transition information, and it does so at a much higher sensitivity than DSC measurements [3], the value of information derived from DMA far exceeds the sole value of glass transition.
The storage and loss modulus measurements are essential for evaluating the mechanical performance of materials, particularly in response to environmental conditions such as temperature and stress. More importantly, for a formulation chemist, understanding how a component utilized in the formulation impacts responsive behavior can enlighten the formulator to avoid either a specific pathway that could spell disaster if employed in non-suitable applications or entertain ideas that can meet the challenges of demands and deliver a solution to the market at a faster pace. The analysis that can be performed using DMA goes far beyond the ultimate property testing when attempting to achieve "failure mode" analysis in other material property testing, as it can reveal the conditions whereby the material can exhibit weakness over the various test parameters of the equipment. The DMA testing aims to study responses to low forces that do not destroy the material's structure. Subtleties of material properties associated with energy responses that cannot be observed in other material test methods can be easily revealed using DMA. When varying a coating's composition or comparing different coating types, the DMA testing can determine:
- Material flexibility, ensuring adequate protection and longevity.
- A material's stability over various temperatures, providing long-term durability and performance insights.
- The degree of crosslinking in coatings.
DMA Instrumentation and Techniques[4]
DMA instruments can be designed for different testing modes, including tensile, bending, shear, and compression; however, the testing attempts to understand material responses when altering one or more of these variables:
- Temperature: DMA can be performed over various temperatures, evaluating coating performance from low to high temperatures.
- Frequency: Testing at different frequencies helps understand how coatings behave under various loading conditions.
- Strain Amplitude: Strain control allows for assessing the material's response to varying mechanical stress.
For this effort, each sample combination maintained a similar stoichiometric index. Samples were prepared as free-film samples and were allowed to cure under ambient conditions for more than 30 days before evaluation. The samples were then tested in tensile mode over the temperature range of -40 °C to 180 °C at a temperature ramp of 3 °C per minute with a strain-controlled analysis at 1 Hz. The data was collected using a TA Instrument Q800 Instrument.
Polyaspartic Chemistry
As discussed earlier, polyaspartic ester chemistry was introduced into the coatings arena in the early 1990s and opened a world of possibilities with the potential to achieve lower VOCs in coatings [5]. In addition to the benefit of lower VOCs, the technology also offered quick setting characteristics. Unlike pure polyurea, which reacts extremely fast and is almost impossible to utilize without specialized dispensing equipment, the polyaspartic technology blocks the amine compounds (also known as hindered amines) to slow down this family of polyurea chemistry and allow for a broadened market reach.
Over 30 years, many have worked to find the optimal combination of components in an aspartic formulation to meet the aspects of various applications — whether to slow down the reaction speed, modify the sensitivity to moisture, or impart a unique characteristic to withstand the conditions of use. The study presented within this paper focuses on the chemical structure of components and their impact on the final coating.
For this study, the components have been categorized based on their unique structure, as detailed below in Table 1.
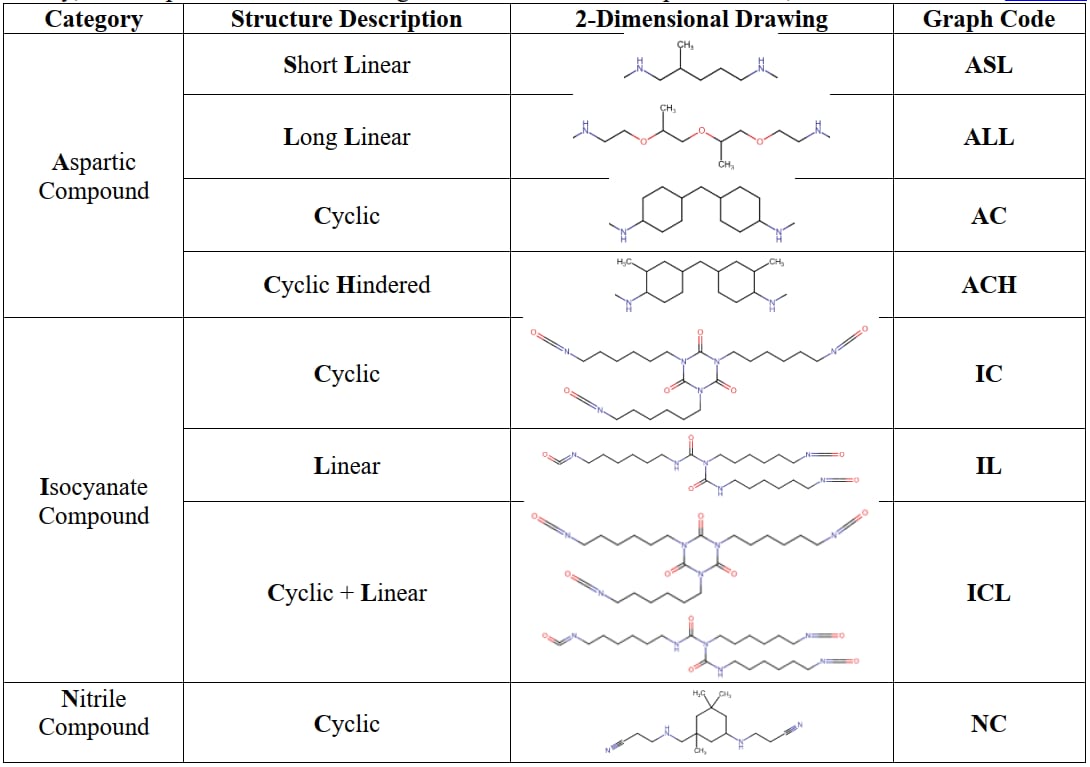
Table 1: Structural representation of compounds employed in the study.
Results and Discussion
Introductory Charts
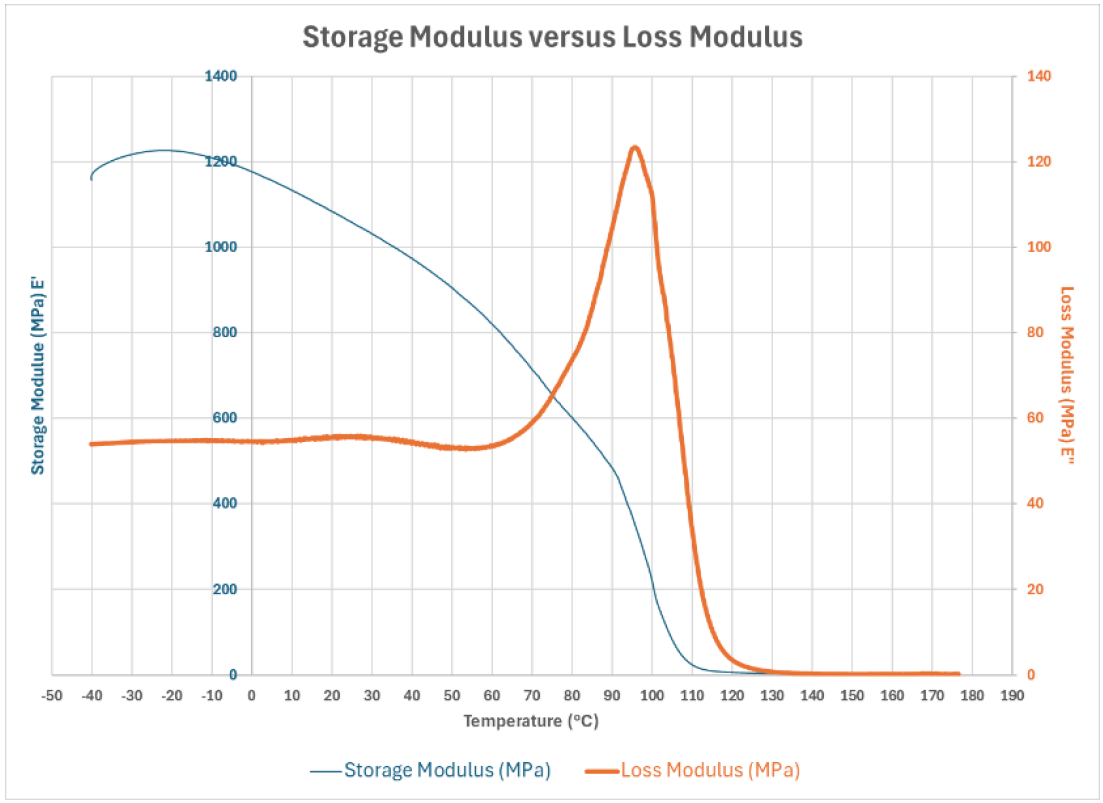
To begin the journey of revealing the impact of compounds evaluated in this study on a polyaspartic system, let’s first start by detailing the concepts of the data obtained by DMA. Stiffness is a term most can easily associate; therefore, Graph 1 illustrates the relationship between the less common term Storage Modulus and Stiffness. As demonstrated in the chart, while the units utilized differ between the two values (N/m for Stiffness and MPa for Storage Modulus), the relationship between these values is similar – the material’s stiffness or storage modulus drops as temperature increases.
Graph 1: Storage Modulus (E’) versus Stiffness.
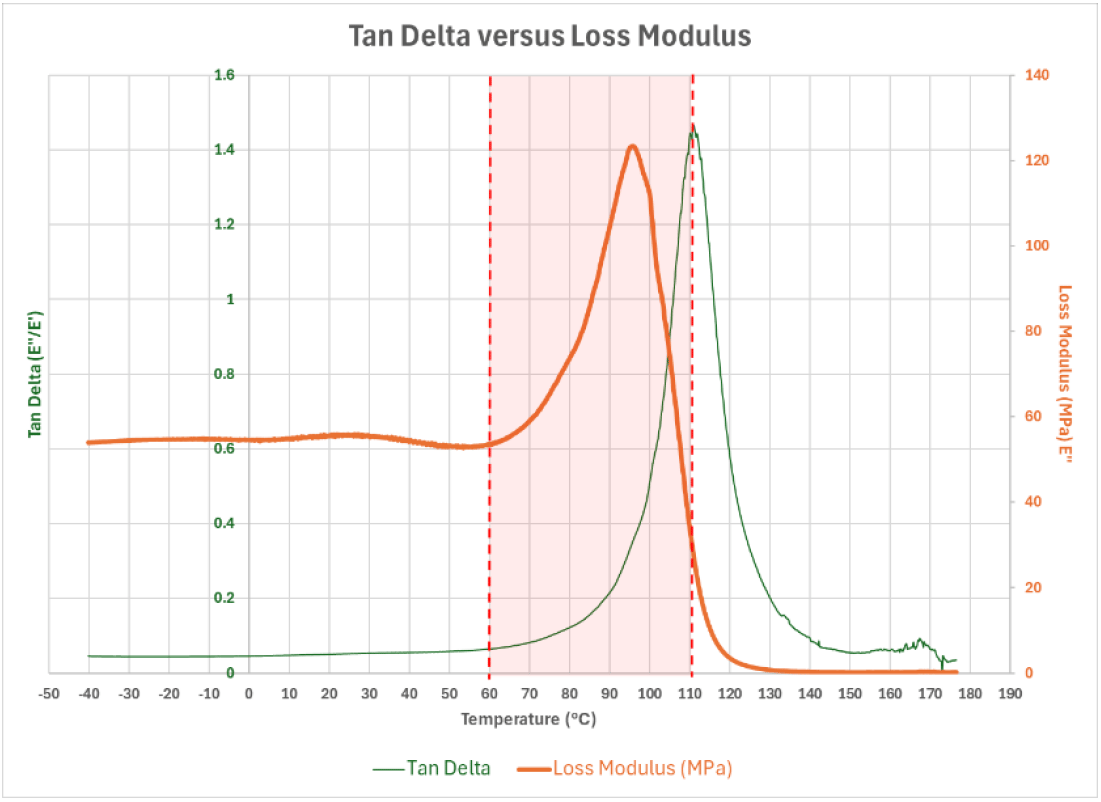
Graph 3: Tan Delta (E''/E') versus Loss Modulus (E')
Moving on to Graph 2, the material’s dissipation of energy, i.e., Loss Modulus (E”), increases as the Storage Modulus (E’) drops, and both behaviors of decreased Storage Modulus and increased Loss Modulus occur as temperature increases. In other words, the material becomes less rigid and more pliable as the temperature increases. Then, in Graph 3, which illustrates the comparison of Tan Delta (E”/E’) to Loss Modulus (E”), the glass transition range starts at 60 °C and reaches 110 °C when ultimate deformation is observed. While each of the three graphs (Graph 1 through Graph 3) illustrates the responses of the material over the entire test parameter on temperature (-40 °C to 180 °C), many will want to evaluate the material’s behavior for the anticipated material’s usage for the given application. As is repeated often in the coatings industry, "There are no bad coatings; only bad applications." This statement derives from the fact that every coating has a window of suitable and non-suitable uses. The material used for Graphs 1 through 3 for illustration purposes would not be appropriate for an application where temperatures exceed 110 °C and may be limited in specific applications above 60 °C.
Composition Comparison Charts
We will now explore how the various compounds identified in Table 1 influence a polyaspartic system on viscoelastic behavior using data collected from the DMA and illustrated in graphs. Graph 4 illustrates the influence of various Isocyanate Compounds (IL – Linear, IC – Cyclic, ICL – Combination of both IL and IC) on the viscoelastic behavior when combined with the cyclic aspartic compound notated in Table 1. The data reveal that the linear isocyanate structure improves both the stiffness of the material along with the ability to dissipate energy over the cyclic structure; however, the use of both the linear and cyclic structure of the isocyanate with the cyclic aspartic compound exposes a synergistic effect, standing out in both an ability to have improved stiffness and ability to dissipate energy from forces.
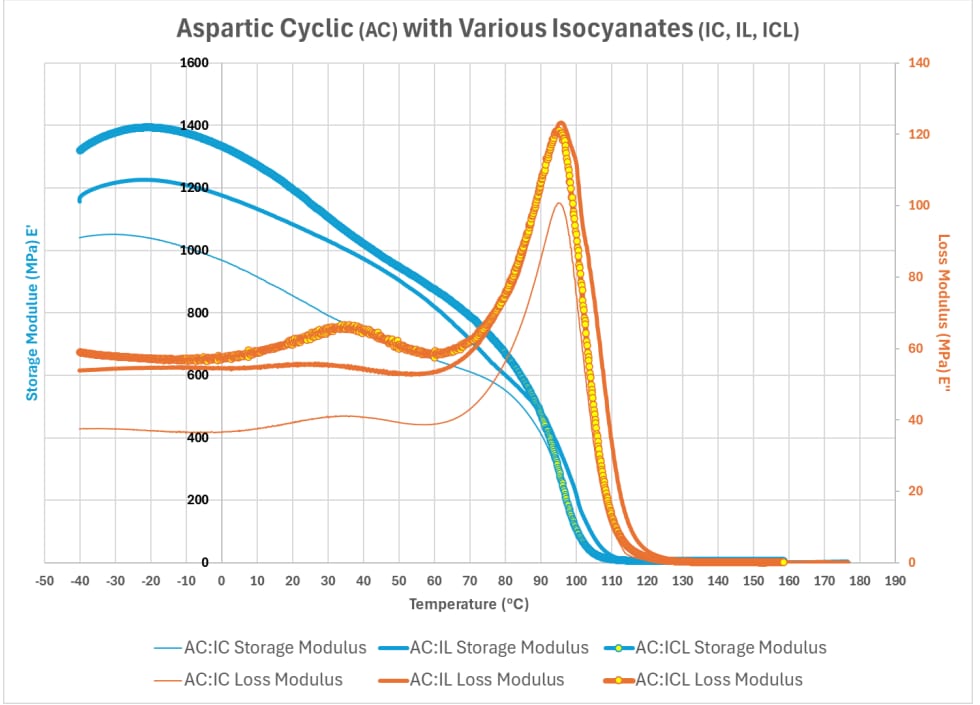
Graph 4: Aspartic Compound (AC) with Various Isocyanate Compounds (IC, IL, ICL)
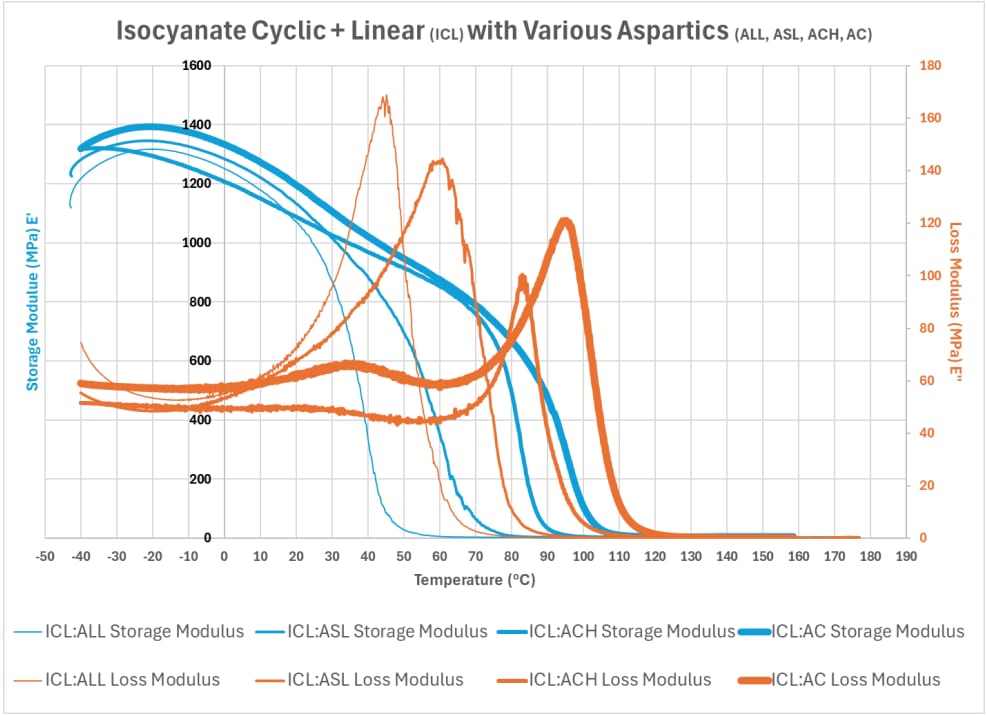
Graph 5 illustrates the results of combining the cyclic and linear isocyanate compound (ICL) with the varying aspartic compounds identified in Table 1: long linear (ALL), short linear (ASL), cyclic hindered (ACH), and cyclic (AC).
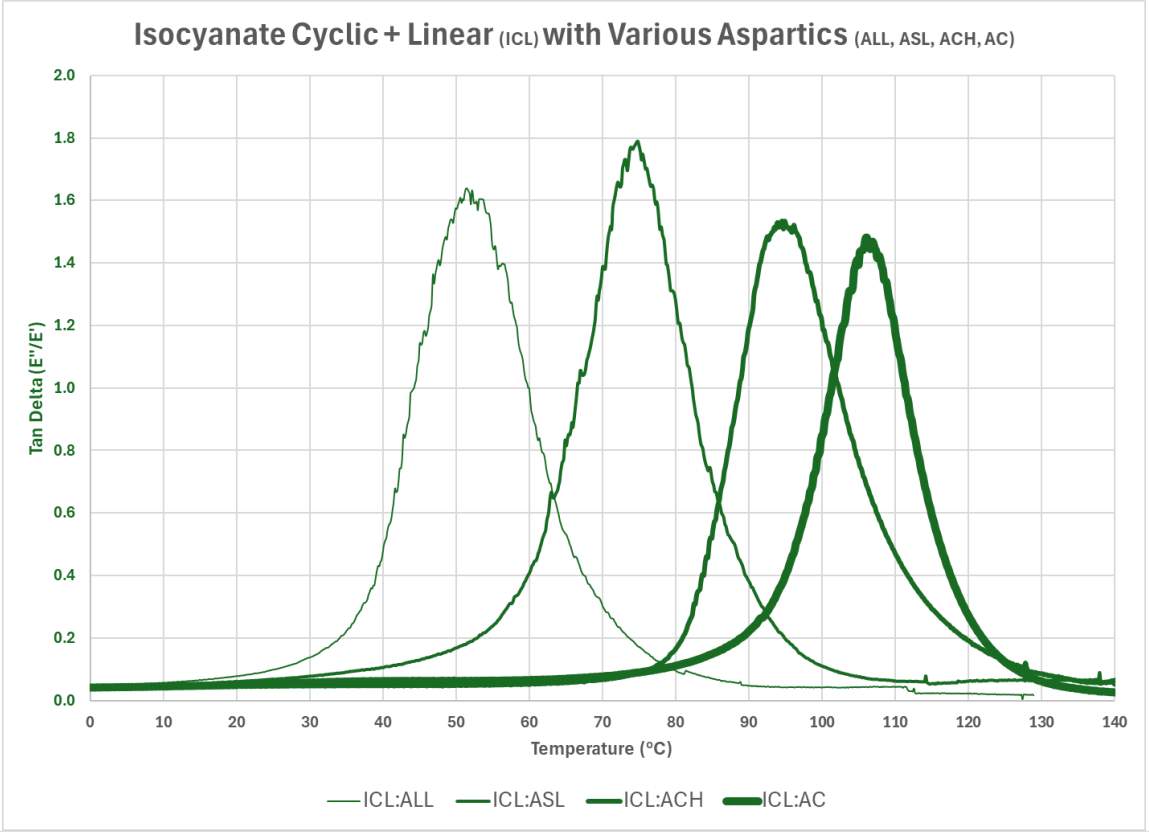
Graph 5: Isocyanate Cyclic and Linear Compound (ICL) with Various Aspartic Compounds (ALL, ASL, ACH, AC)
Using DMA to evaluate the influence of modifying the aspartic compounds with the varying structures, Graph 5 demonstrates that adding linear structures impacts the storage and loss modulus behavior with lower temperature exposures. It can be more clearly illustrated using the Tan Delta information, as detailed in Graph 6 and Table 2, below:
Graph 6: Tan Delta of Isocyanate Cyclic and Linear Compound (ICL) with Various Aspartic Compounds (ALL, ASL, ACH, AC)

Table 2: Influence of structure on Tan Delta Peak Temperature.
While the impact of switching from a cyclic aspartic compound to a hindered cyclic aspartic compound had a slight influence on viscoelastic behavior, the art of formulating a coating system lies in combining the correct compounds to impart the final properties needed for a given application. Aspartic systems are not different from other systems in that many tools exist in a formulator's tool chest that can influence final formulation properties. Graph 7 illustrates how incorporating a cyclic nitrile compound with the hindered cyclic aspartic compound can benefit the final properties of the system, even exceeding specific properties achieved with the cyclic aspartic compound illustrated in Graphs 5 and 6.
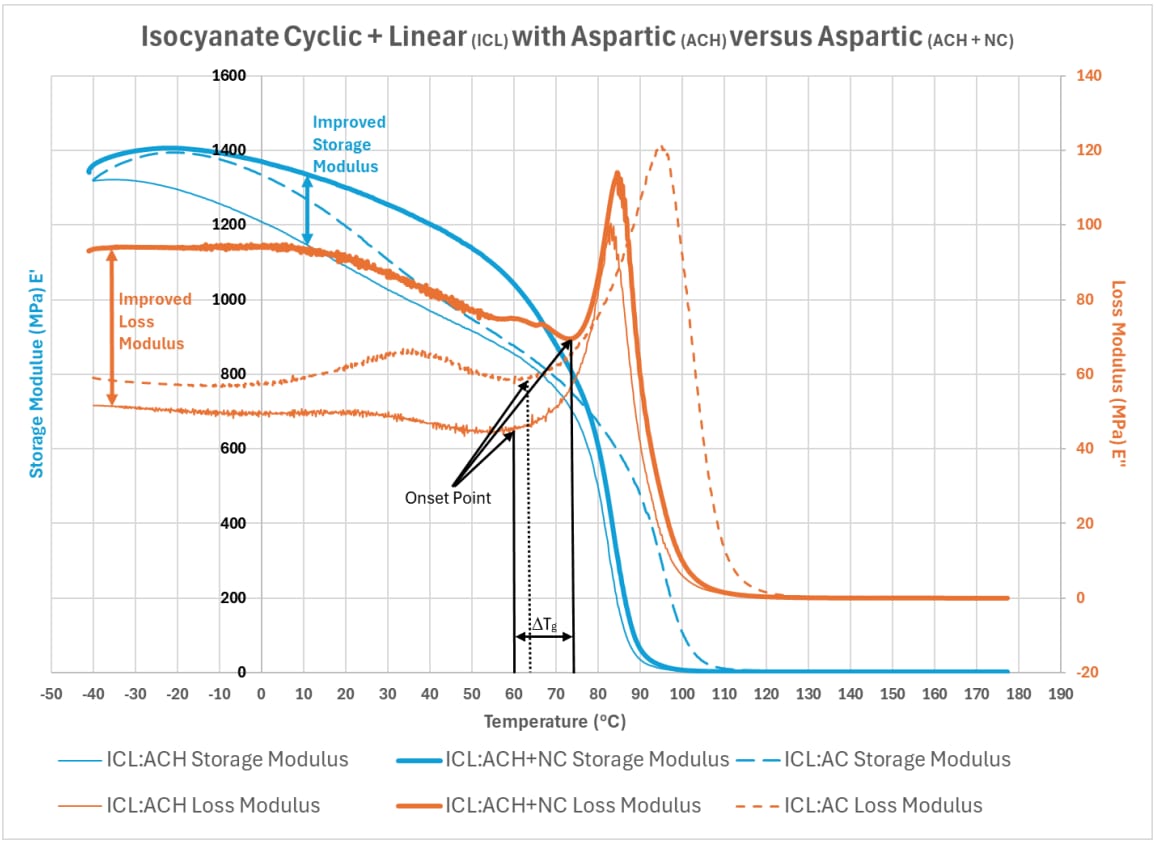
Graph 7: Isocyanate Cyclic and Linear Compound (ICL) with Aspartic Compounds (ACH) versus the Aspartic Compound with a Nitrile Compound (ACH + NC)
The key takeaways from Graph 7 include that the use of the cyclic nitrile compound with the hindered cyclic aspartic compound develops an improved:
- Storage Modulus (E')
- Storage Modulus (E') over the cyclic aspartic compound with a similar isocyanate compound
- Loss Modulus (E")
- Loss Modulus (E") over the cyclic aspartic compound with a similar isocyanate compound
- The onset-point for Loss Modulus (E"), improving the Tg.
- The onset-point for Loss Modulus (E") over the cyclic aspartic compound with a similar isocyanate compound, improving the Tg.
In addition to the above improvements illustrated in Graph 7, the following Tan Delta information is noted:

Table 3: Influence of structure on Tan Delta Peak Temperature.
While the cyclic nitrile compound increases the temperature at which permanent deformation occurs compared to the cyclic hindered aspartic system, it does not reach the Tan Delta peak temperature achieved with the cyclic aspartic compound sample; however, the improved onset-point could open opportunities that would not exist in specific applications with the increased glass transition.
Conclusion
DMA is an effective thermal analysis instrument that captures informative data regarding a material’s viscoelastic behavior. By employing DMA testing, the formulator can optimize the composition to meet the requirements for the desired application. The rapid characterization made possible using DMA can reduce the development process time. This paper illustrated how, by varying the composition of aspartic systems based on differing structural compounds, the DMA could discern the influence of those modifications to guide a formulator in achieving the intended goal for their system design.
Acknowledgements
"Evaluating Modifications in Aspartic Systems on Viscoelastic Behavior" 2025 Polyurethanes Technical Conference 6-8 October 2025, St. Louis MO, USA, Published with permission of CPI, Center for the Polyurethanes Industry, Washington, DC.
References
[1] Coasey, Keith (Ph.D.), “Understanding Biopolymer Behavior.” TA Instruments / Waters Corporation. New Castle, DE. 2024.
[2] Dunson, Debra (Ph.D.), “Characterization of Polymers using Dynamic Mechanical Analysis (DMA).” EAG Laboratories. Columbia, MO. Rev. 06, 2017.
[3] Foreman, J. Saurebrunn, S.R., Marcozzi, C.L. “Exploring the Sensitivity of Thermal Analysis Techniques to the Glass Transition. (TA082)” TA Instruments / Waters Corporation. New Castle, DE.
[4] Franck, A, “Introduction to Dynamic Mechanical Analysis and its Application to Testing of Polymer Solids. (TA441)” TA Instruments / Waters Corporation. New Castle, DE.
[5] Pflaumer Brothers, Inc. “An Introduction to Polyaspartic Coatings.” https://pflaumer.com/an-introduction-to-polyaspartic-coatings Accessed 24 January 2025.
Opening image courtesy of Vadym Terelyuk / iStock / Getty Images Plus.
